
Metal displacement occurs when a more reactive metal forms a cation and displaces a less reactive metal from its compounds.ĥ. This provides the driving force for metal displacement reaction. In other words, a highly reactive metal prefers to be a cation while its less reactive friend prefers to be a free element. A less reactive metal is a hoarder - it would rather have its valence electrons. The reactivity series tells us how much a metal wants to be a cationĪ more reactive metal donates electrons more readily to form a cation. More reactive metals react more vigorously, increasing the rate of effervescence of hydrogen gas.Ĥ. Secondly, reactivity affects the rate of reaction. Reactive nonmetals are a group of chemical elements that are located in the upper right-hand corner of the periodic table.This group includes 11 elements, namely hydrogen (H), carbon (C), nitrogen (N), oxygen (O), fluorine (F), phosphorus (P), sulfur (S), chlorine (Cl), selenium (Se), bromine (Br), and iodine (I). The students will also be interacting with their journals using INB templates for periodic table and reactivity. The periodic table and reactivity lesson plan includes a PowerPoint with activities scattered throughout to keep the students engaged. The alkali metals, found in group 1 of the periodic table, are highly reactive metals that do not occur freely in nature. Watch the YouTube video Reaction (Explosion) of Alkali Metals with Water. For our purposes we will define the following ten families: Alkali metals. The unreactive metals do not react at all, even when we use strong acid or high temperature. The Periodic table can also be divided into several families of elements each having similar properties.Moderately reactive metals require a higher temperature to react with water.Highly reactive metals are trigger happy, reacting with acids and even water at room temperature.Reactive metal + hydrochloric acid ⟶ metal chloride + hydrogenįirstly, reactivity affects what a metal can react with. Reactive metal + steam ⟶ metal oxide + hydrogen 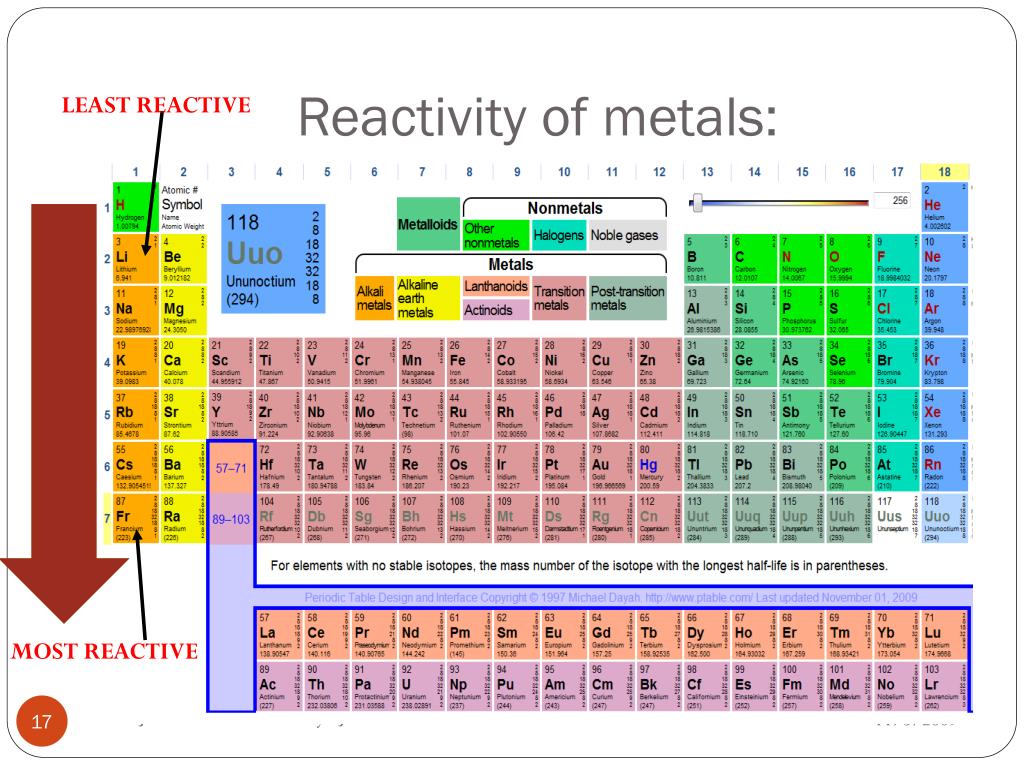
Reactive metal + water ⟶ metal hydroxide + hydrogen During a single-displacement reaction, a metal higher. At the bottom are the least reactive metals. At the top of the list are the highly reactive metals that lose electrons during a chemical reaction to form ions. The largest atomic radius of elements in their period. The reactivity series tells us if a reaction will happen and under what conditions Metal The reactivity series of metals is a list of metals arranged in their order of reactivity from highest to lowest. Highly reactive, with reactivity increasing moving down the group. The trio sit in the same column within the transition metal hood.ģ. The Trio of Unreactive MetalsĪnd the least reactive metals we need to know are copper, silver, and gold. Lead in Group IV is also moderately reactive. Transition metals are generally less reactive than the main group metals. Within each group, the metal lower down the group is more reactive. They have the greatest tendency to lose electrons to form cations.įor the four metals we need to know, those from Group I are more reactive than those from Group II. Group I alkali metals and Group II alkaline earth metals are the most reactive. The reactivity series is written into the Periodic Table! Highly Reactive Group I and II Metals
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
